“Our work suggests a potential risk of SARS-CoV re-emergence from viruses currently circulating in bat populations.”
These were words written by Professor Ralph Baric’s group in 2015, foreshadowing a pandemic that would define a generation in just a few short years. It would have benefited all of us to heed these prophetic words more closely.
In the final weeks of 2019, the city of Wuhan, China reported an outbreak of atypical pneumonia. The patients were administered antibiotics, and their health did not improve, leading the attending physicians to believe that it was likely virus-driven and not bacteria. The initially reported cases were linked to a seafood market in Wuhan, prompting many to speculate that the infectious agent was acquired there. Additionally, given that the cases came from the same wet market location on the same date, the human-to-human transmission was not believed to have been the cause of the outbreak. Instead, this appeared to be a spillover event of a virus into a new host.
Spillover occurs when a virus that has maintained transmission in a particular species infects a new one; it “spills over” into a novel host. The majority of spillover events are dead ends. The virus could infect a novel host but is not capable of sustaining a transmission cycle amongst that new host. This happens with high frequency, particularly in areas where human and zoonotic populations overlap. Avian influenza is an excellent example of this. High pathogenicity avian influenza strains such as H5N1 and H7N9 have repeatedly spilled over into human hosts. Thankfully the current strains of high pathogenicity avian influenza are self-limiting, and human-to-human transmission is extremely limited or non-existent.
However, viruses adapt. Occasionally a virus will acquire enough of the right mutations to allow it to infect a novel host and allow for continuous transmission in said host. The human immunodeficiency virus (HIV) is an excellent example of this. HIV did not simply snap into existence; it resulted from a spillover from the simian immunodeficiency virus, a virus circulating in non-human primates for tens of thousands of years. It is believed that, in the early 20th century, handling of bushmeat from an infected primate and blood-to-blood contact with the handler occurred, resulting in the initial spillover event that eventually led to the AIDS pandemic.
Coronaviruses are quite common in humans. Some strains are responsible for the common cold and viral pneumonia. In 2002, the severe acute respiratory syndrome coronavirus (SARS) burst onto the scene. Unlike most other known coronaviruses, SARS was a very aggressive and lethal virus, with a case-fatality rate near 10%. Symptoms included fever, shortness of breath, an opacity of the lungs upon chest x-ray, and a curious drop in lymphocytes (white blood cells) after infection. Being a virus, the infection was ambivalent to antibiotic treatment. Human-to-human transmission occurred, particularly in healthcare settings where nurses and physicians treating infected patients became ill themselves. The international alarm of the new virus led to major travel restrictions, quarantining of entire hospital staff, and the moving of the 2003 Women’s World Cup Tournament.
By the middle of 2004, SARS had seemingly disappeared. The international crisis was averted, but not before nearly 800 people succumbed to the virus. The work of where this virus originated from and its spread had already begun with reports that palm civets could carry the SARS virus, but they didn’t appear to be the major host organism of SARS. A few years later, however, came the wake-up call: bats were acting as major reservoirs of a multitude of what we now refer to as SARS-related coronaviruses. As the years progressed, we began to appreciate just how prevalent these viruses were in bats and how their prevalence was contributing to their evolution.
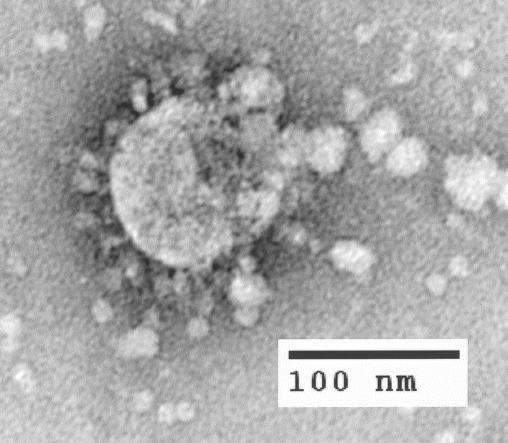
Coronaviruses are given their name through the crown-like appearance of the virus particle under electron microscopy (corona is Latin for “crown”). The crown appearance is due to the Spike protein encoded by the virus, which binds to host cell receptors known as angiotensin-converting enzyme 2 (ACE2), mediating viral cell entry. All coronaviruses are encoded by an RNA genome, in contrast to humans, which use DNA. The replication of RNA is usually more error-prone than DNA, and as such, RNA-encoded viruses typically display higher mutation rates than their DNA counterparts, such as herpesviruses. However, coronaviruses add an extra layer of evolutionary complexity in that multiple coronavirus variants can infect the same bat. This allows for a process called recombination, where coronaviruses can swap genetic material within the same host.
A single bat can carry multiple distinct SARS-related coronaviruses. Inside of the bat, the individual viruses can propagate and be secreted, sustaining a transmission cycle. However, the simultaneously present SARS-related coronaviruses can also swap genetic material, a process called “recombination”. This allows for the generation of novel, recombined SARS-related coronaviruses to be produced.
Take the example below. A single bat carries two SARS-related coronaviruses, which we are terming “SARS-related Coronavirus A” (in red) and “SARS-related Coronavirus B” (in blue). As the separate viruses replicate inside of the bat, pieces from Virus A and Virus B can swap out, generating recombinant SARS-related coronaviruses (in purple). The overwhelming majority of times that this occurs, it is bad for the virus. Chunks of genetic material being interchanged in a relatively small genome will usually lead to recombinant viruses with either non-functional proteins or viruses with some fitness disadvantages. But still the original SARS-related coronavirus A and SARS-related Coronavirus B will live on, and continue to propagate themselves inside a new bat where the process can begin again. Perhaps this time a recombinant virus will not have any survival disadvantages. This is how the original SARS is believed to have come into existence: through the recombination of multiple distinct coronaviruses hosted in bats.
It took scientists roughly 13 years to track down where the original SARS came from, and it will likely take a few years for us to trace down the events that lead to the emergence of SARS-CoV-2. All of the current evidence suggests that bats were the original host reservoir and that the virus eventually spilled over into humans. It is quite likely that the direct ancestor of SARS-CoV-2 has been circulating in bats for decades. Keep in mind that the simian immunodeficiency virus, the ancestor of HIV, was infecting non-human primates long before it spilled over into humans. In another example, the Rabies virus, another RNA virus, has an intrinsically high mutation rate in the face of strong selective pressures. Rabies virus is a member of the lyssavirus genus and exists as “ecological ensembles,” metapopulations of distinct species and variants, residing within multi-reservoir mammalian communities in often rapidly changing environments. Bats, one of the mammalian reservoirs for lyssaviruses, allow for rabies strains to be globally mobile with ongoing mutation for change and adaptation.
As our understanding of SARS-CoV-2 has progressed, we remain skeptical that the seafood market was the location of the initial spillover of SARS-CoV-2 into the human population. Other cases not linked to the wet market around the same time in Wuhan were later identified, lending support that the spillover event had occurred earlier. This would translate to the wet seafood market, not as the spillover site but rather the first known superspreader site. From what we know about how SARS-CoV-2 is transmitted, a densely-packed location such as a wet seafood market would be ideal for a burst of new infections. Just like a Rose Garden ceremony at the White House or a church choir event.
Just one year ago, the first known cluster was identified in Wuhan. Since then, SARS-CoV-2 has spread to every continent in the globe, including Antarctica. Continuous adaptation of the virus to its new human host has been observed throughout the pandemic, from the D614G variant in the spring/summer to the B117 variant in the United Kingdom and South Africa in recent weeks. Although less lethal than its cousins SARS and MERS, SARS-CoV-2 has proven itself to be the single greatest challenge to public health we have seen in decades and perhaps a century.
So where are we today? Simply put, SARS-related coronaviruses present one of the greatest threats to future pandemics. The original SARS emerged in 2002, the related MERS coronavirus emerged in 2012, and finally, SARS-CoV-2 in 2019. The identification of coronaviruses with high pandemic potential and lethality are becoming the norm rather than the anomaly. The continuous transmission cycles of these coronaviruses in zoonotic populations represent a massive public health concern due to the: (i) increasingly overlapping locations of zoonotic hosts and humans, (ii) increased urbanization in these locations, and (iii) the ability for coronaviruses to swap genetic material within their zoonotic hosts, giving rise to swarms of new recombinants.
The speed at which this respiratory pathogen torched its way through the world is stunning. It certainly received an assist from domestic and international fumbling of public health communications. We’re not stating that SARS-CoV-2 is behind us; much to the contrary, we believe that SARS-CoV-2 will be with us for the foreseeable future. Even in the most optimistic scenarios for vaccine effectiveness, rollout, and uptake, we will continue to see tens of thousands of hospitalizations due to COVID-19 well into the late spring of 2021. Instead, we propose the need to recalibrate our focus on emerging infectious diseases and seek dedicated, line-item eternally funded public health funding and support much as we do for other domestic and foreign threats via Department of Defense funding. We must understand that microbes pose an equal or greater threat to our health and well-being than that of our wartime or terrorist threats. We believe they are even more dangerous because they are invisible and are capable of adapting to counter our efforts. The unspeakable acts of violence and domestic terrorism at the Capitol building on January 6 claimed the lives of at least five people That same day, there were at least 3,844 COVID-19 associated deaths in the United States.
Surveillance of SARS-related coronaviruses in zoonotic populations has never been more critical. Indeed, public health surveillance and control efforts are paramount to our collective global “One Health.” While our ongoing fight against SARS-CoV-2 continues, other SARS-related coronaviruses continuously circulate amongst bats. Most of these viruses are either incapable of infecting humans or will likely be self-limiting as a spillover event would not lead to subsequent infections (i.e. a dead-end). However, just like picking a lock, the continuous evolution of SARS-related coronaviruses gives them many attempts to find the right combination.
There’s going to be a SARS-Coronavirus 3.
______________________________________________________________________________________________________________________






